
B6: Role of interferon regulated factor (IRF) 4 for Th17 differentiation

Prof. Dr. Michael Lohoff
Institut für Medizinische Mikrobiologie
und Krankenhaushygiene
Hans-Meerwein Straße 2
D-35032 Marburg
Phone: +49- 6421-58 64345
Fax: +49-6421-58 66420
E-Mail: M. Lohoff
Homepage: Medmikrobio
Cooperation within LOEWE
B5 - Burchert
C4 - U. Bauer
B2 - Stiewe
B4 - Suske
Graduate student
Nadine Bollig
Email: N. Bollig
Personal advisory board: Lohoff, Burchert, Konur
| B6: Role of interferon regulated factor (IRF) 4 for Th17 differentiation |
|
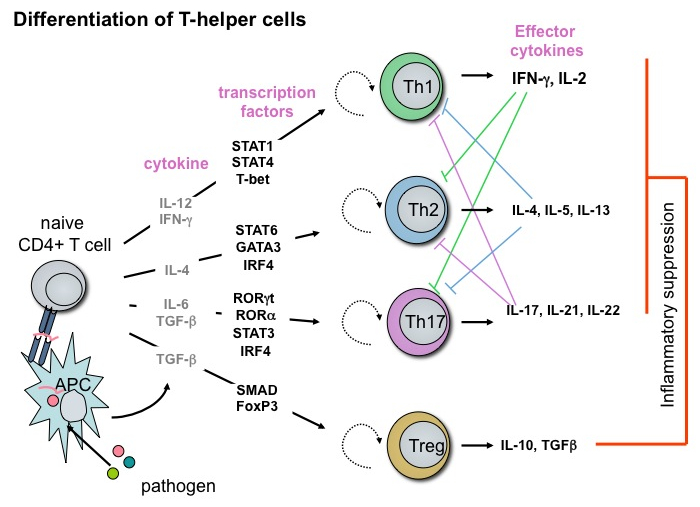
CD4+ T cells can be classified into Th1- Th2-, Th17- and regulatory T (Treg) cells according to their specific cytokine profile upon stimulation (see also corresponding figure). During differentiation from naive CD4+ cells to the different subgroups, cytokine-induced specific transcription factors play a dominant role. Our group was the first one who could demonstrate that Interferon-regulator-factor (IRF) transcription factors are responsible for the formation of the different T cell subtypes. IRF1 and IRF2 are important for Th1 differentiation and play also a dominant role during murine leishmaniosis. IRF4 was identified as the ‚master-transcription factor’ for Th2 development and its absence is associated with a dramatically enhanced activation-induced cell death (AICD). IRF4 is the central transcription factor not only for Th2- but also for Th17-differentiation with significance also for the development of experimental autoimmune encephalomyelitis (EAE), the murine disease model for multiple sclerosis (MS).
Project description
Beside STAT3 and RORγt, IRF4 is the central transcription factor for Th17 differentiation, albeit IRF4 is expressed by all CD4+ subtypes upon T-cell activation. IRF4-expression by itself is therefore not sufficient to direct T-cell differentiation into a particular subtype, but additional IRF4-binding-‚partners’ are necessary and/or modifications at the transcription factor itself. A still open question is also how IRF4 induces Th17 differentiation in the presence of TGF-ß plus IL-6, but does not play any role for Treg differention in the presence of TGF-ß alone.
The central question to be answered in our project is therefore to specify the role of IRF4 during Th17 differentiation. Chromatin precipitation of IRF4 and ChIP-Seq technique will allow to define IRF4-binding sites genomewide.
Further we will also analyse the impact of the different transcription factors and their mutual interaction during T cell differentiation at different time points. For this we will establish an inducible retroviral Tet-On-system. Hereby we will able to answer the following question:
Our findings will also allow to analyse the significance of the tumor microenvironment on the generation of T cell subpopulations with pro- and/or anti-tumoral properties.
 |
| Last Updated on Monday, 07 December 2009 16:16 |

