Research
| Research |
|
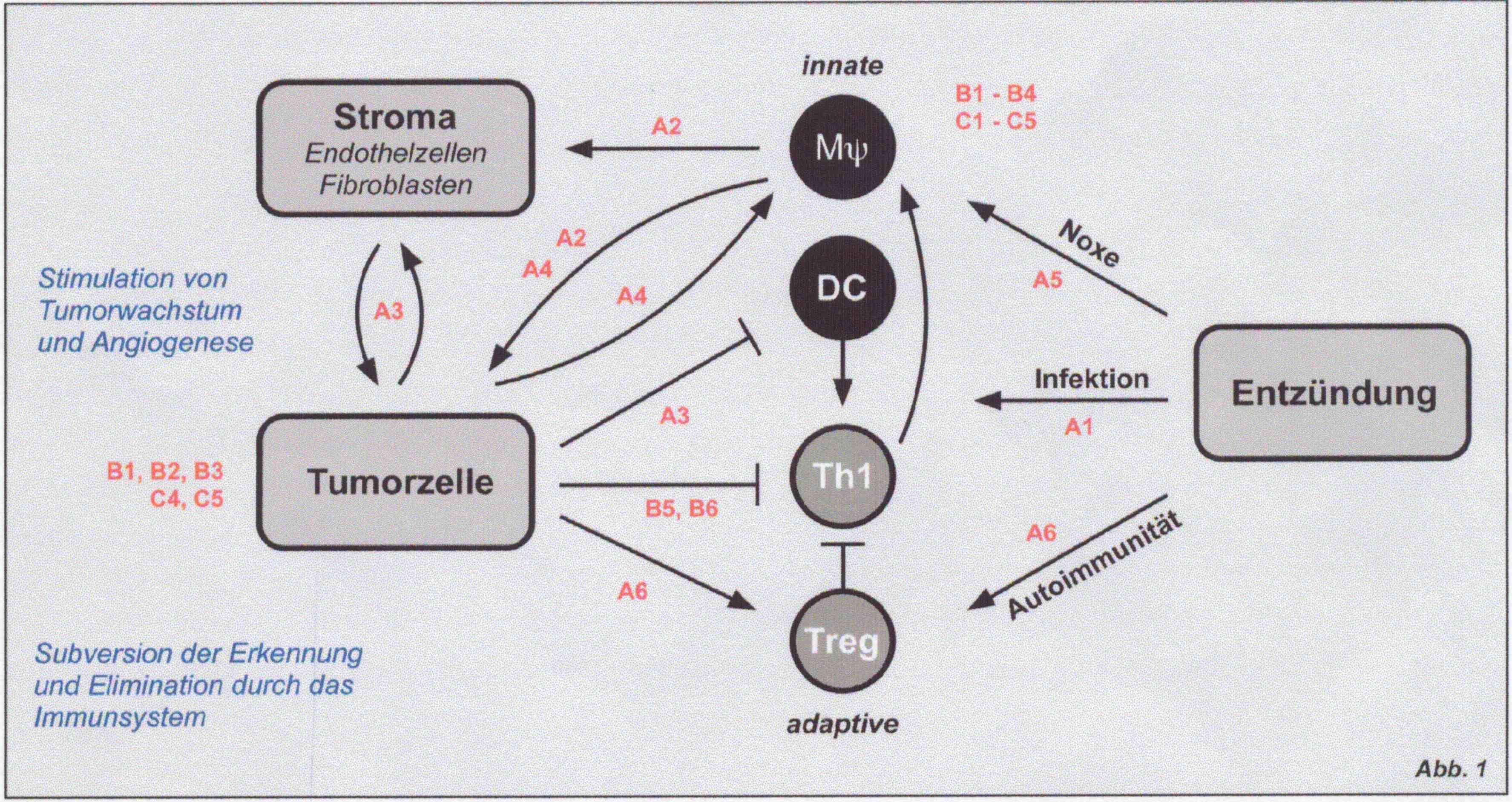
Progress over the past years in the field of tumor- and inflammation research highlighted the strong overlap between both research areas. Particularly the fact that the same intracellular signalling cascades and the same transcription factors are used by both, tumor- and immune cells respectively, illustrates the mechanistic similarity in both cell types. Indeed, inflammation and tumor progression are often positively correlated e.g. in colon and pancreas carcinomas. Therefore, it is of particular interest to elucidate further these mechanistic similarities between tumor- and immune cells and to investigate in detail their mutual impact on each other. A deeper knowledge of the similarities and differences within the signaling cascade (ranging from receptor/ligand interaction to transcription factors and to epigenetic mechanisms as chromatin modification and remodeling) will allow for therapeutic intervention strategies.
The LOEWE research program is divided into three synergistic work packages with the joining intention to elucidate the significance of tumor-, stroma- and inflammatory cell interactions (e.g. macrophages, endothelial cells, fibroblasts and lymphocytes) and their impact on tumor formation and progression. The focus is on the transcriptional signalling pathways and on epigenetically modulated regulation mechanisms supporting tumor progression and neoangiogenesis. Research program A: Cellular interaction and cytokine induced signalling pathways This research program focuses on intracellular signalling pathways induced by mediators like interleukines and interferones during intercellular mutual interaction of tumor- and inflammatory cells. Results from recent work demonstrate that not only cytokines but also neuropeptides and neurotrophines have a significant impact on intracellular signalling mechanisms.
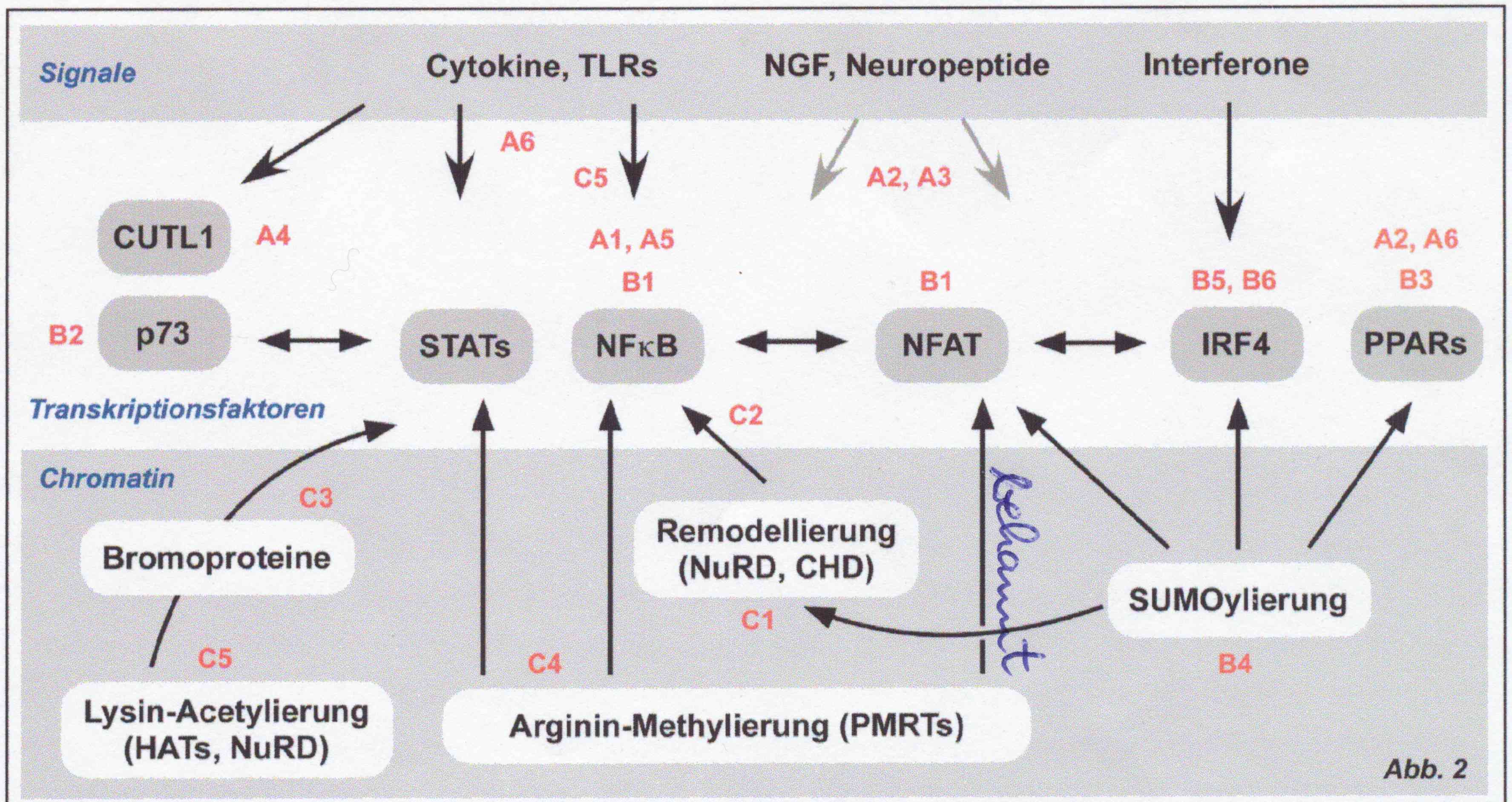
Research program B: Regulation of transcription factors A key event upon triggering of tumor- as well as immune cells by mediators e.g. cytokines, bacterial components (toll like receptor (TLR) ligands), interferones and neuropetides is a dramatic alteration in the gene expression profile of target cells induced by the induction and/or modulation of specific transcription factors. The research program B will mainly focuse on those transcription factors with an outstanding significance for tumor- and immune cell responses like NF-kB, STATs, NFAT, IRFs and PPARs.
Research program C: Epigenetically modulated regulation mechanisms Induction of gene expression profiles is not only modulated by direct modification of transcription factors (e.g. by phosphorylation), but also by epigenetic changes, in tumors as well as in inflammatory cells. Mechanisms for the dynamic changes of the chromatin structure are the histone-modification (e.G. lysine- acetylation and methylation, arginine methylation) and the involvement of ATP-dependent chromatin remodeler. The focus of this research program is to resolve the mutual interaction of these two mechanisms and their significance for specific gene expression in tumor and inflammation.
|
| Last Updated on Tuesday, 14 December 2010 10:10 |